In the dense tropical rainforests of Central and South America, leafcutter ants have perfected an agricultural system that rivals human farming in complexity. These tiny farmers cultivate fungal gardens, their primary food source, with remarkable precision. Recent research reveals a fascinating parallel between their practices and modern human agriculture: the use of antibiotic rotation to combat resistant pathogens. This discovery not only sheds light on the sophisticated strategies of these insects but also offers potential insights for addressing one of humanity's most pressing medical challenges—antibiotic resistance.
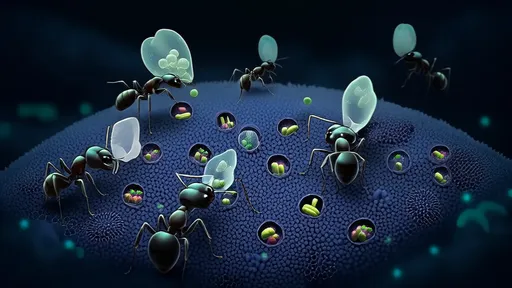
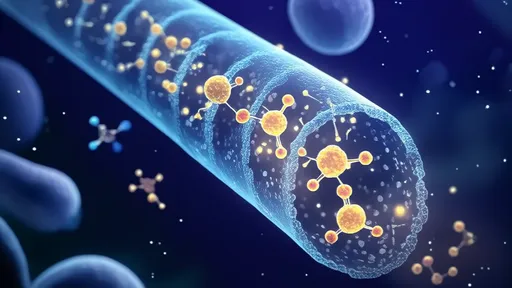
The leafcutter ants' fungal gardens are vulnerable to invasion by a parasitic fungus called Escovopsis, which can devastate their crops. To protect their food supply, the ants employ a multi-layered defense system. They carry antibiotic-producing bacteria on their bodies, which secrete compounds that suppress the growth of Escovopsis. What researchers find particularly intriguing is that these ants appear to alternate between different strains of bacteria, effectively rotating their "antibiotics" to prevent the parasite from developing resistance.
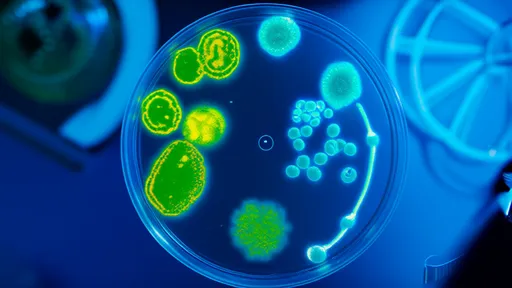
This phenomenon mirrors the concept of antibiotic rotation in human medicine, where clinicians alternate between different classes of antibiotics to slow the emergence of resistant bacterial strains. The ants' system, however, has been refined over millions of years of evolution. Their bacterial partners produce a diverse array of antimicrobial compounds, and the ants seem to modulate their use based on the threat level posed by Escovopsis. When the parasitic fungus becomes more aggressive, the ants respond by increasing the diversity of their microbial defenses.
Scientists studying this system have identified several key lessons that could inform human approaches to antibiotic resistance. First, the ants maintain a diverse "pharmacopoeia" of antimicrobial compounds, which allows them to switch between treatments as needed. Second, they apply these compounds proactively, before infections become established, rather than waiting for outbreaks to occur. Third, their system is decentralized—each ant colony maintains its own unique blend of bacterial symbionts, creating a constantly shifting landscape of antimicrobial defenses that pathogens struggle to overcome.
The implications for human medicine are profound. Our current approach to antibiotics often involves using single compounds in high doses until resistance emerges, at which point we scramble to develop new drugs. The ants' strategy suggests that maintaining a diverse arsenal of antimicrobials and rotating their use systematically might be more effective in the long term. Some researchers are now exploring whether similar principles could be applied in clinical settings, perhaps by developing cocktails of multiple antibiotics or by alternating between natural antimicrobial products.
Beyond antibiotic rotation, the leafcutter ants' farming system offers other insights into sustainable agriculture. Their fungal gardens are remarkably efficient at breaking down plant material, a process that human biofuel researchers are keen to understand. The ants also practice waste management, removing infected garden material and depositing it in special refuse piles away from their nests. This hygienic behavior helps prevent the spread of disease, much like crop rotation and field sanitation in human agriculture.
As we face growing challenges from drug-resistant infections and the need for more sustainable agricultural practices, nature's solutions—perfected over eons of evolution—may provide valuable guidance. The leafcutter ants' sophisticated farming techniques, developed without human-style technology or centralized planning, demonstrate that complex problems can sometimes be solved through decentralized, adaptive systems. Their antibiotic rotation strategy, in particular, offers a compelling model for how we might approach our own struggle against resistant pathogens.
Ongoing research continues to uncover new details about this remarkable symbiotic system. Scientists are now sequencing the genomes of the ants' bacterial partners to identify novel antimicrobial compounds that could be developed into human medicines. Others are studying how the ants regulate their microbial defenses, looking for clues about how we might better manage our own use of antibiotics. What began as curiosity about some of nature's most dedicated farmers has blossomed into a rich field of study with potentially transformative applications for human health and agriculture.
The story of leafcutter ants and their fungal gardens reminds us that some of the most innovative solutions to our problems may already exist in nature. By observing and learning from these tiny agricultural experts, we may find ways to strengthen our own defenses against the rising tide of antibiotic resistance while developing more sustainable approaches to feeding our growing population. As we confront the limitations of our current medical and agricultural paradigms, the ants' millions of years of evolutionary wisdom offer both inspiration and practical guidance for the path forward.
By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025